CENTRALIZED MPS HUB: UNDER ONE ROOF FOR MULTI‑PLATFORM PRECLINICAL SOLUTIONS.
 Kidney
Kidney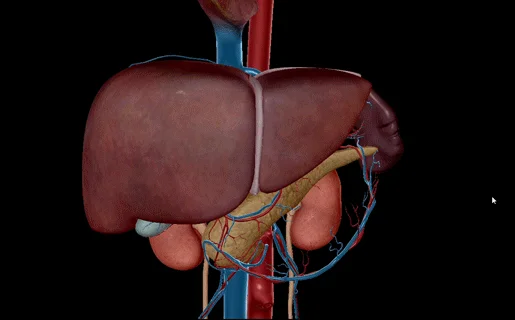 Liver
Liver Immune
Immune GI
GI Ocular
Ocular Repro
Repro Neuro
Neuro CV
CVWe focus where it matters, integrating traditional tools with innovative NAMs through strategic partnerships and in-house development to drive human-relevant insights in biology, efficacy, safety, and ADME.